They began to be held in 1911 and brought together the greatest scientists of the moment, among them were practically all those who gave their contribution to the current atomic model.\] They met at the famous conferences sponsored by Ernest Solvay, a Belgian-born chemist and industrialist, who became famous in the world of science. It seems incredible, but most of the physicists who contributed to the current atomic model appear in the same photo. Influential scientists on the current atomic model In this way, spin becomes the fourth quantum number. So the same state characterized by the quantum numbers n, l, m of the Schrödinger equation, can be occupied by at most 2 electrons with opposite spins.Spin does not appear in Schrödinger's equation, but is incorporated into the atomic model through the Pauli principle: The electron is a fermion with two possible spin states +½ and -½.The square of the wave function is the probability density of finding the electron at a given position, viewed from the center of the atomic nucleus. The wave function determines the regions allowed for the electron.The equation of the current model has a solution for some discrete values of energy, so that the famous Planck quanta appear naturally from the solutions of the Schrödinger equation.In the case of many-electron atoms, the orbitals of the outermost electrons are modeled by the potential of the nucleus shielded by the innermost electrons, which is known as the Debye potential.
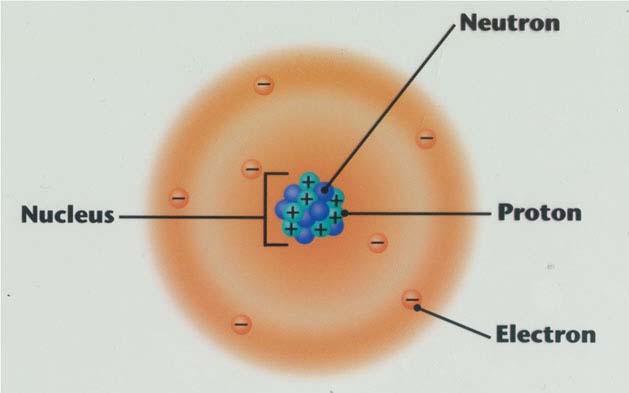

The nucleus would be like an ant in the center of the field, but with a staggering 99.9% atomic mass. To get a scale picture of the current atomic model, let's imagine that an atom has a diameter like that of a soccer field. The mathematical square of the wave function determines the orbitals, that is, the areas around the nucleus where the electrons can be found with greater probability. The Schrödinger wave equation predicts which values of energy and angular momentum are allowed, as well as the wave function associated with each level of energy and momentum. And when they interact with matter atoms, they have particle behavior.ĭue to this, the electrons around the nucleus are scattered in zones called atomic orbitals, whose shape and extent depend on the energy level of the electrons and the angular momentum.īoth the energy and the angular momentum of the electron around the nucleus have certain allowed values, therefore they are said to be quantized. On the other hand, with photons, the opposite happens, in most optical phenomena (at the macroscopic level) they have a fundamentally wave behavior. In the current atomic model, the behavior of the electron at the scale of atoms is prominently wavelike, while at the macroscopic level, like the electrons moving in the cathode ray tubes of old televisions, corpuscular behavior predominates. This is Louis De Broglie's famous wave-particle duality. The rest is an image far from reality, since the scale of the nucleus does not even correspond to that of the atom: the nucleus is 1/100,000 times the size of the atom, but that is where the atomic mass is concentrated.Ĭlassical mechanics states that every material particle has an associated wave, called the wave function. The classical image is useful to see that the nucleus contains two protons and two neutrons and guaranteeing the neutrality of the atom there are two electrons occupying the same energy level.
